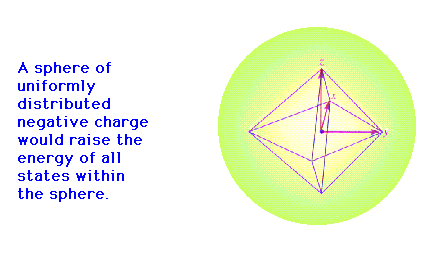
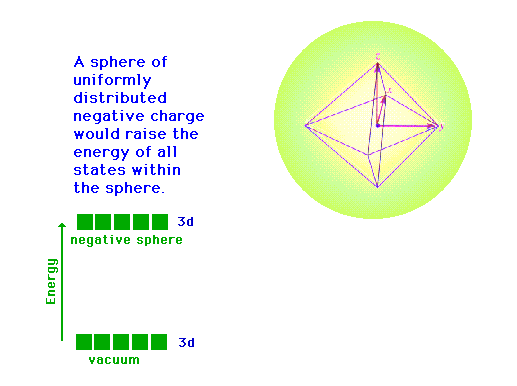
| CURMUDGEON GENERAL'S WARNING. These "slides" represent highlights from lecture and are neither complete nor meant to replace lecture. It is advised not to use these as a reliable means to replace missed lecture material. Do so at risk to healthy academic performance in 09-105. |
Structural Isomers
Stereoisomers
Geometrical isomers
Square planar geometries
Octahedral geometries
Optical isomers
Observations to explain
Colors
Magnetic Properties
Stability
Crystal Field Theory