| Lecture
#20, October 18, 2006 |
Cruising in to
Chapter 16
|
| CURMUDGEON
GENERAL'S WARNING. These "slides"
represent highlights from lecture and are neither
complete nor meant to replace lecture. It is
advised not to use
these as a reliable means to replace missed
lecture material. Do so at risk to healthy
academic performance in 09-105. |
|
| Lecture Outline (some material shown is
look-and-listen only) |
Quantum Theory of the Chemical Bond
Molecular orbitals (in "homonuclear diatomic
molecules"
Approximated by combinations of atomic
orbitals
Constructive and
destructive interference effects
Combining 1s atomic
orbitals
"Sigma"
bonding molecular orbital
"Sigma"
antibonding molecular orbital
|
| If destructive interference represents the orbital in
which the electron happens to be found, the linear
combination results in a depeletion of electron density
between the positively charged protons. They repel each
other under these circumstances. |
 |
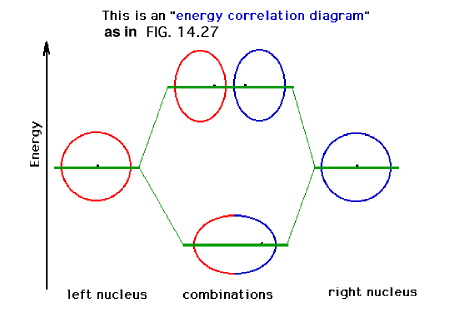
| A schematic of the orbitals on separated protons
relative to "close" protons. The constructive
combination of atomic orbitals gives rise to a system
lower in energy than the isolated system and is a bonding
molecular orbital. |
 |
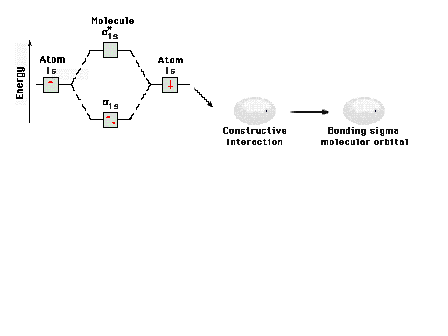
| Energy diagram and geometry for the sigma-1s
(bonding) orbital construction. |
 |
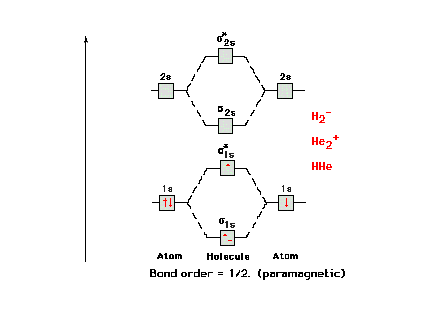
| Re-defining how one calculates bond order within the
context of molecular orbital theory. |
 |
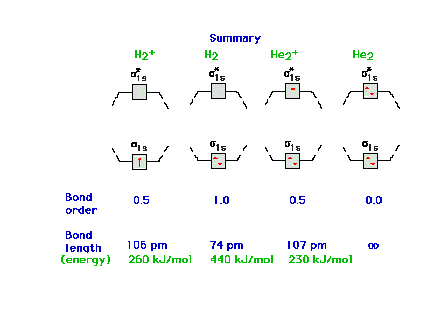
The molecular orbital energy diagram for H2-
and other isoelectronic species, all having bond order =
0.5. The electron configuration would be written as s1s2 s*1s2
s2s2
or, as in the current text, simply ss2 s*s2
ss2. |
 |
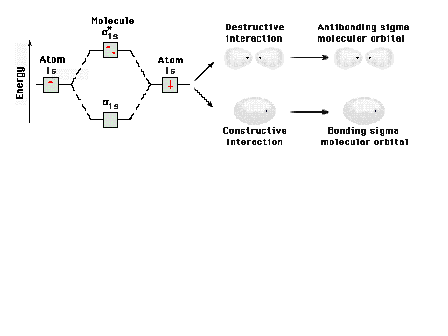
| Energy diagram and geometry for the sigma*-1s
(antibonding) orbital construction |
 |
| Molecular orbital energy diagrams for 1, 2, 3, and 4
electrons systems. The correspondence between bond order
and bondlength and between bond order and bond energy is
shown for each molecular species. |
 |
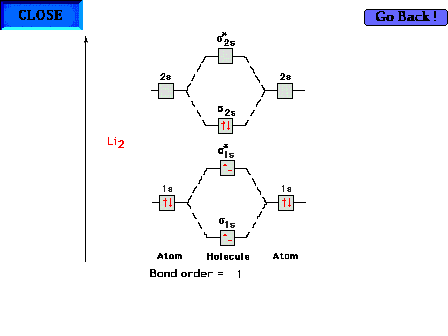
| The molecular orbital energy diagram for Li2
containing all six electrons. Since antibonding electrons
negate the effect of bonding electrons, the pair of s* electrons cancels out
the bonding characteristics of the pair of s electrons and both pairs revert to
the inner core 1s2 configurations that we
expect from simpler considerations. The electron
configuration could thus be written as [He][He]s2s2. |
 |
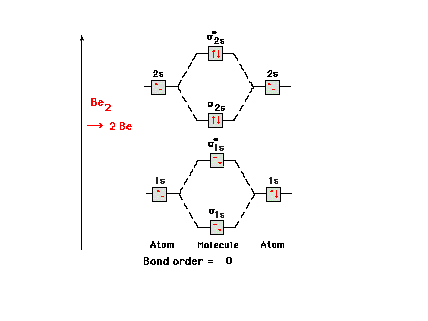
| The molecular orbital energy diagram for Be2.
Since the number of antibonding electrons is equal to the
number of bonding electrons, there is no net bonding in
this molecule and it simply breaks back up into two Be
atoms. |
 |
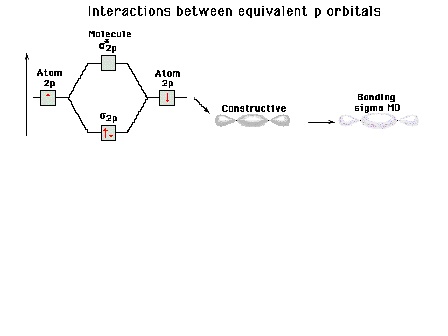
| Energy diagram and geometry for the sigma-2px
(bonding) orbital construction from the 2p atomic
orbitals originally directed along the bonding (x) axis. |
 |
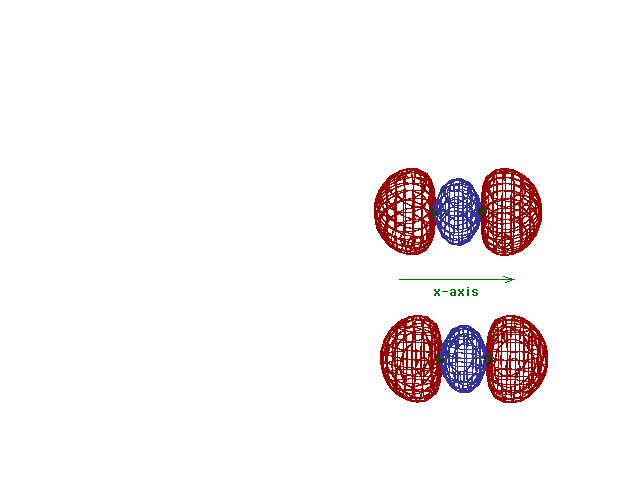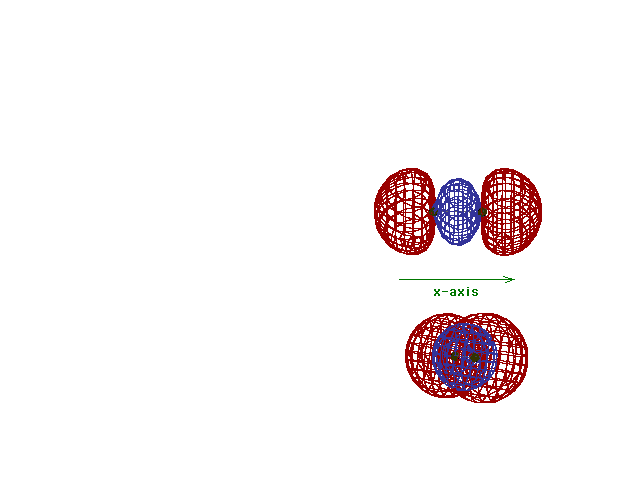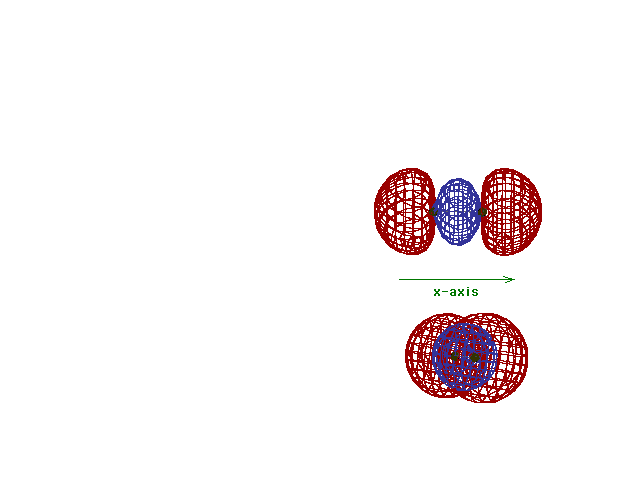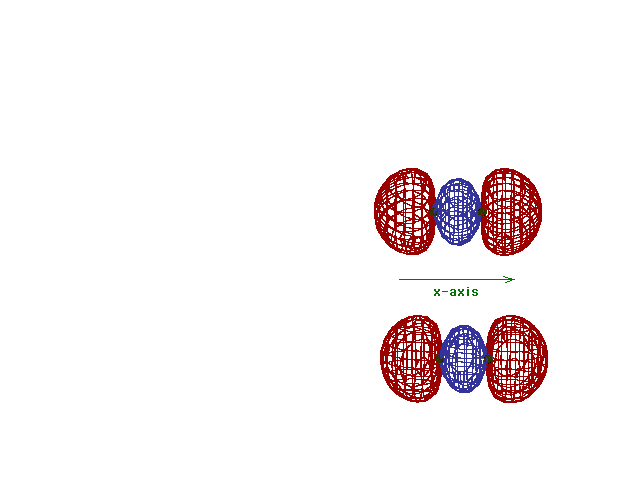
| This (with luck) is an animation of the
computer-generated s2p
(or sp) bonding
molecular orbital showing how the symmetry about the bond
axis resembles that for the s-atomic orbital's symmetry
about the nucleus. Hence the s label. |
 |
| Energy diagram and geometry for the sigma*-2p
(antibonding) molecular orbital construction |
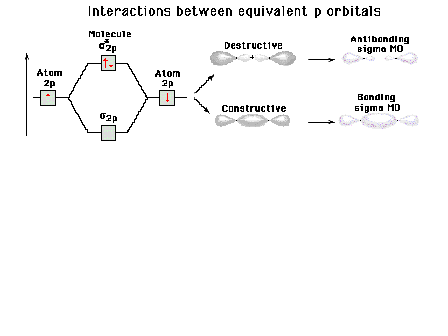 |
| A reminder that, perpendicular to the bond axis in
the x-direction, there are 2p atomic orbitals in the
z-direction and in the y-direction. Just the 2py
orbitals on each atom are shown here. |
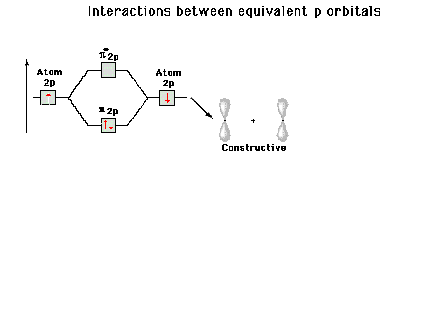 |
| Energy diagram and geometry for the pi-2py
(bonding) molecular orbital construction from the 2p
atomic orbitals originally pointing in the y-direction,
perpendicular to the bond axis and in the plane of the
illustration. Constructive interference occurs in the
region of overlap. (Recognize that there is an identical
pi bonding level, same energy, constructed in the
x-direction from the 2pz atomic orbitals.) |
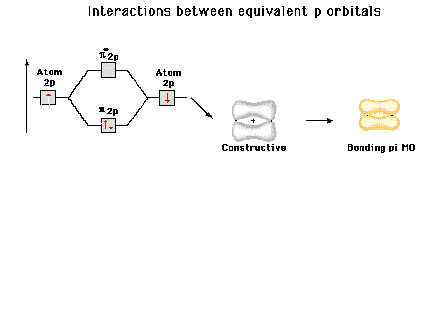 |
| For "diboron", B2 we get a
somewhat surprising result out of the molecular orbital
energy diagram and the ensuing electron configuration.
The ninth and tenth electrons go into the lowest energy
available orbitals. These are the p orbitals. There are two of equal
energy and Hund's rule forces us to put one electron in
each and with parallel spins. Thus we have bond order =
1, a single bond, but it is not a sigma bond. In fact,
there is one electron in one pi-orbital and another
electron in a second pi-orbital comprising this single
bond. |
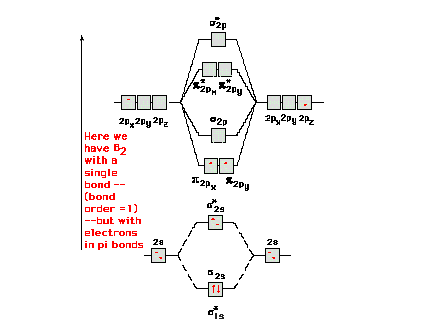 |
