| Lecture
#22 |
| |
| CURMUDGEON
GENERAL'S WARNING. These "slides"
represent highlights from lecture and are neither
complete nor meant to replace lecture. It is
advised not to use
these as a reliable means to replace missed
lecture material. Do so at risk to healthy
academic performance in 09-105. |
|
| Lecture Outline |
Quantum Theory of the Chemical Bond
Molecular Orbitals
Polyatomic molecules
Hybrid atomic orbitals
|
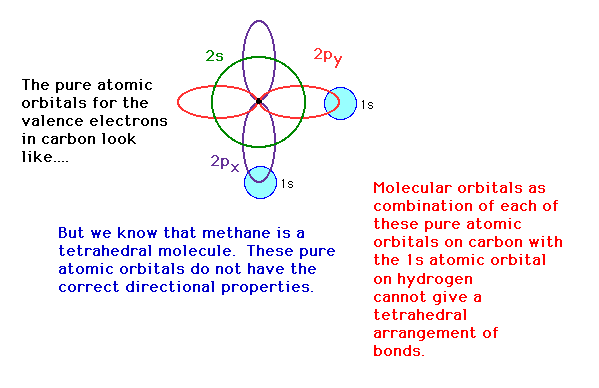
| Polyatomic molecues. In constructing a
valence bond picture for methane, the valence electron
configuration on carbon, 2s22p2
will not return the correct directional properties for
the bonds produced by overlap with hydrogen's 1s atomic
orbitals.
|
 |
| The 2s, 2px, 2py and 2pz
atomic orbital geometries are not tetrahedrally directed.
We need orbitals that have the correct directional
properties and we can synthesize these from our known
atomic orbitals by mixing, a process known here as hybridization.
We will look at an example that the text refers to for
describing the bonding in BeH2. |
 |
| In the BeH2 molecule (HBeH), we need two
atomic orbitals that are directed linearly, according to
the "known" geometry. (VSEPR tells us this.)
Constructing hybrid orbitals from the 2s and 2px
atomic orbitals by taking appropriate mixtures. These are
being shown for illustrative purposes. The math is not
part of the required material. "2s" represents
the pure 2s atomic orbital wave function, for example.
The numerical coefficients are not crucial to the
demonstration. |
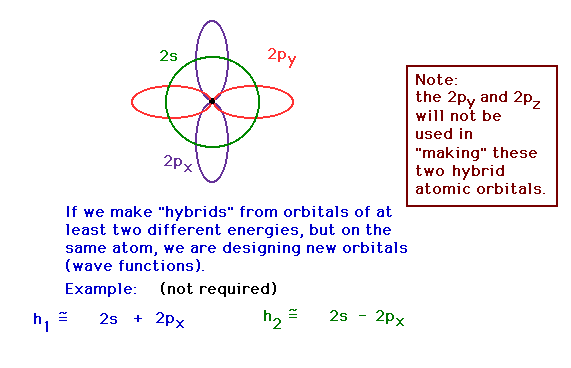 |
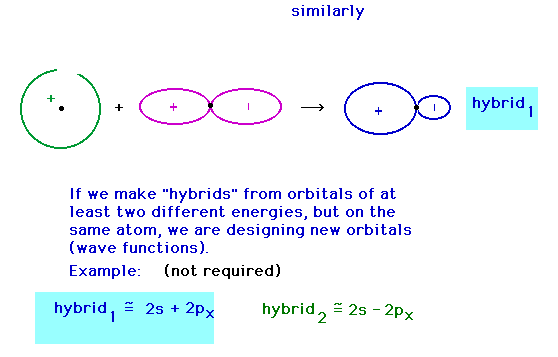
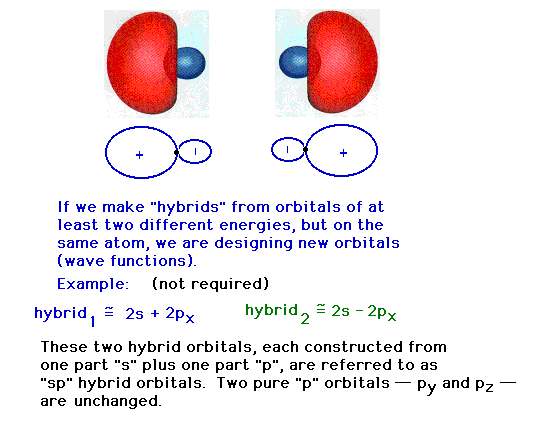
| Hybridizing from the 2s and 2px atomic
orbitals giving a hybrid orbital pointing in the
x-direction. |
 |
| Hybridizing from the 2s and 2px atomic
orbitals giving a hybrid pointing at 180o to
the previous hybrid. |
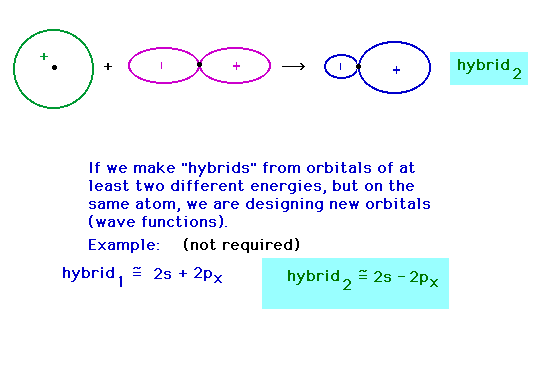 |
| The two pure atomic orbitals we have used have been
re-assembled into two mixed, hybrid atomic orbitals. Each
of the latter are identical in shape, but pointed in
different directions. The remaining 2p atomic orbitals
that have not been used remain unchanged in subsequent
discussions about the full set of valence atomic orbitals
in this system (that is, on the Be in beryllium hydride).
Now we return to a discussion of the atomic orbitals
that are appropriate to a tetrahedral geometry about a
central atom that had pure s and p atomic orbitals when
nothing else was around.
|
 |
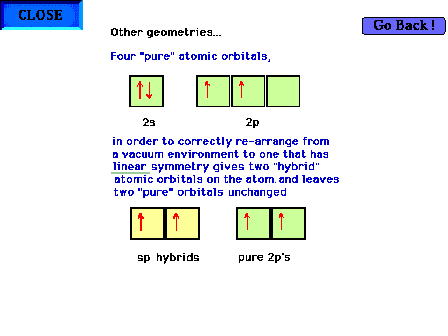
| Hybridization of s and p valence orbitals in a linear
environment (two perturbing
sites). Two of the orbitals,
the s and one of the p orbitals are mixed forming two sp hybrid orbitals, leaving
the remaining two p orbitals
as pure atomic orbitals. |
 |
| Hybridization of s and p valence orbitals in a
tetrahedral environment. That is, the carbon in this case
is surrounded by a tetrahedral arrangment of (four) objects (hydrogens) which
influence the behavior of carbons valence electrons. The
net result of all the forces (carbon at the center and a
tetrahedral arrangement of perturbations) gives four orbitals that are identical
in shape to each other and that are directed to the
vertices of a tetrahedron. These orbitals can be
simulated by hybridizing the four
previous pure orbitals through mixing mathematically and
are each symbolized as sp3 hybrid orbitals. |
 |
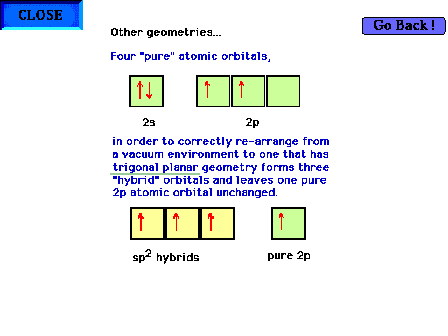
| Hybridization of s and p valence orbitals in a
trigonal planar environment (three
perturbing sites around the central atom). Three valence orbitals, the s and
two of the p's, are hybridized to give three sp2 hybrid
orbitals, leaving one pure
atomic p orbital unchanged. |
 |
| Summary of the effect of various perturbing
geometries on the s/p valence shells in a central atom.
By "vacuum" is meant the atom in isolation from
any external influence. |
 |
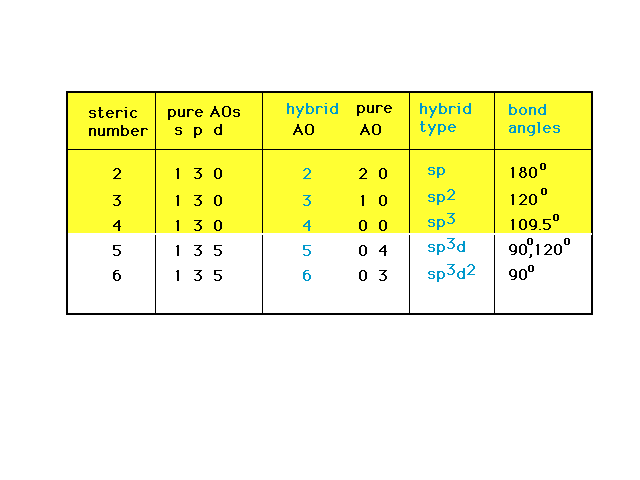
| For expanded octets, the d orbitals come into
consideration. There are five d orbitals (since d implies
the angular momentum quantum number l = 2 and hence ml
= -2, -1, 0, +1, +2. The trigonal bipyramid and
octahedral geometries involve five and
six hybrids as indicated. |
 |
| An overview of the relationship among steric
number, and the number and types of pure and hybrid
orbitals associated with the implied geometries. |
 |
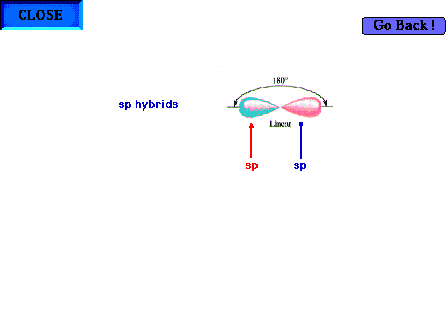
| An illustration of a central atom's two sp hybrid
orbitals present for a linear geometry about the central
atom. Bear in mind that it is implied that there are also
two pure pure p orbitals (not shown) perpendicular to the
indicated axis. |
 |
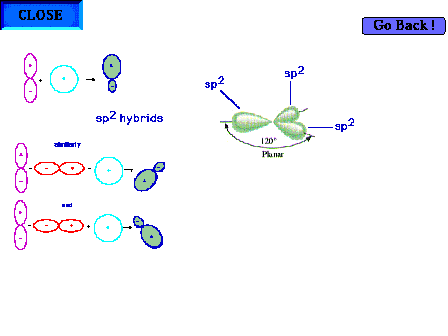
| Three sp2 hybrid atomic orbitals,
constructed in some detail on an earlier slide, showing
their trigonal planar directionality. It is implied that
there is one remaining pure p atomic orbital
perpendicular to the plane shown. |
 |
| Bonding considerations in a polyatomic system
illustrated for the molecule ethylene (aka ethene) H2C=CH2.
The Lewis structure leads to a VSEPR expectation that
each carbon is in a trigonal planar geometry which in
turn indicates that there are three sp2 hybrid
atomic orbitals and one pure p atomic orbital at each
carbon. |
 |
| The atomic orbitals for either individual carbon in
ethylene. |
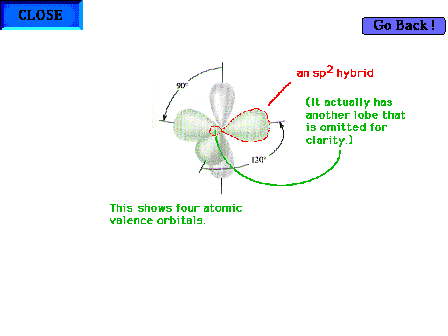 |
| The atomic orbitals for carbon in ethylene
highlighting the one pure 2p atomic orbital
perpendicular to the plane of the three hybrid sp2
orbitals. |
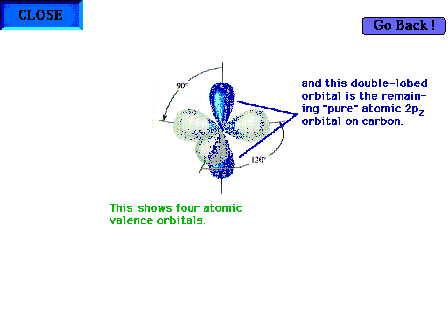 |
| Assembling the ethylene molecule showing the overlap
between the various pairs of atomic orbitals (which can
be pure or hybrid). |
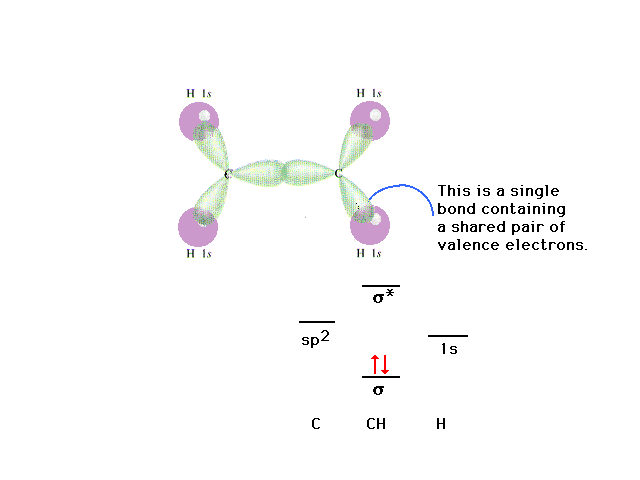 |
| The C-H "sigma" bond. The "sigma"
label comes from the symmetry (appearance) of the
electron distribution about the bond axis. When viewed
directly down the bond axis, the profile resembles the
atomic s-orbital profile. Hence the "sigma"
term. |
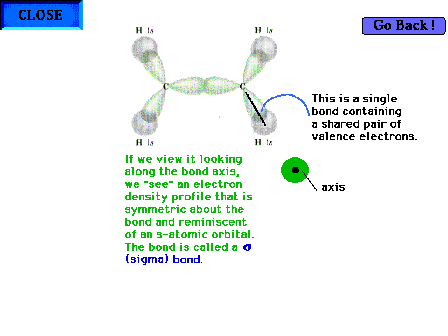 |
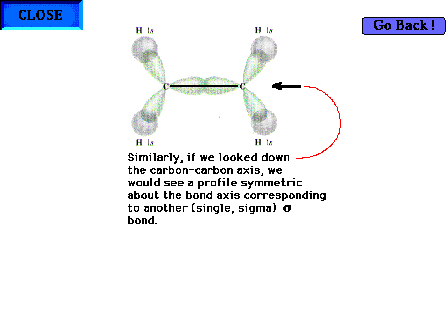
| The C-C "sigma" bond. The profile of the
implied electron distribution about the bond axis is
similar to that on the previous slide for the C-H sigma
bond. |
 |
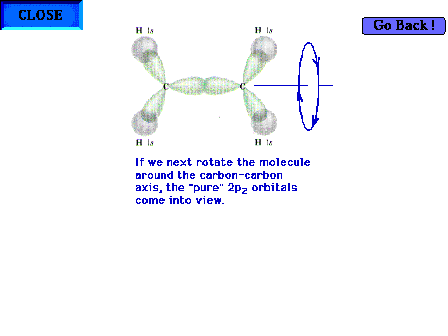
| Rotating the molecule 90o about the C-C
bond axis so that the structure's plane is perpendicular
to the point of view brings the pure 2p atomic orbitals
into view (since they are perpendicular to the sp2
hybrid orbitals). |
 |
| The pair of bars represents one (that's one!)
overlap result from bringing the two neighboring 2p
orbitals closer together. |
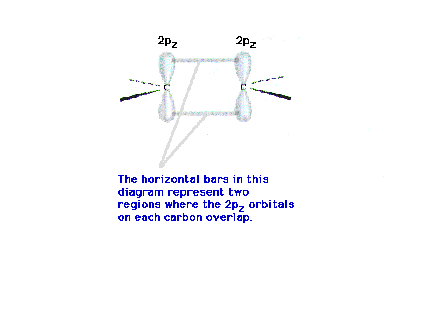 |
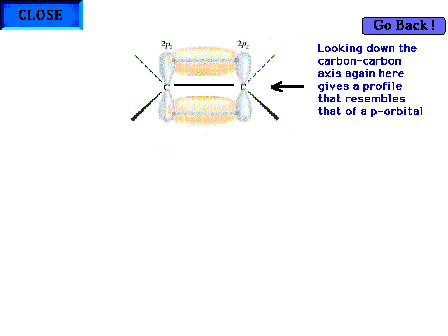
| Looking down the C-C axis again to determine the
geometric appearance of the electron density in this new
region of overlap. |
 |
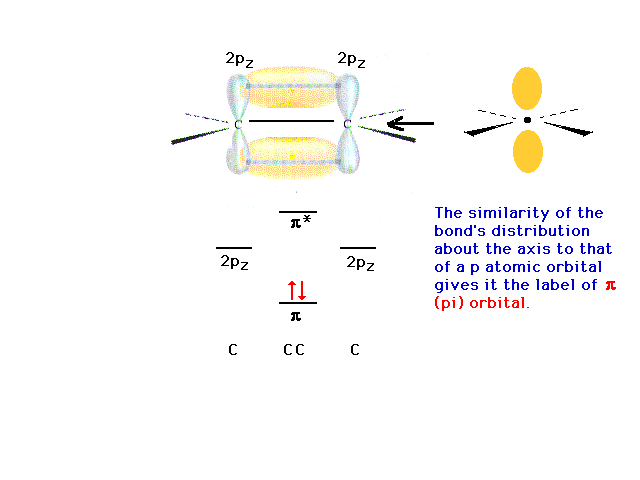
| The view down the C-C axis shows that the electron
density about the axis is analogous to what is seen for
the electron density of a p orbital about a nucleus. We
refer to the bonding density in this arrangement as
constituting a pi bond. |
 |