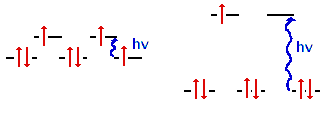CARNEGIE MELLON
UNIVERSITY
09-105 Introduction to
Modern Chemistry
Quiz #9
December 1, 1998
Name
| H |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
2He |
| Li |
Be |
|
|
|
|
|
|
|
|
|
|
B |
C |
N |
O |
F |
10Ne |
| Na |
Mg |
|
|
|
|
|
|
|
|
|
|
Al |
Si |
P |
S |
Cl |
18Ar |
| K |
Ca |
Sc |
Ti |
V |
Cr |
Mn |
Fe |
Co |
Ni |
Cu |
Zn |
Ga |
Ge |
As |
Se |
Br |
36Kr |
A hexadentate ligand is sold by a
chemical manufacturer under the tradename "Bettis." Co2+ forms a hexadentate chelate complex with this
ligand and a solution containing this complex ion appears
colorless. Can you tell from this information whether
"Bettis" is a weak-field ligand or a strong-field
ligand? Explain briefly.
Co2+ has 25 electrons and a
configuration given by [Ar]3d7. The two possible
configurations in an octahedral environment correspond to weak
field and strong field ligands shown here.
The left figure is for
a weak field ligand and shows three unpaired spins.
The right figure is for a strong field ligand and shows
one spin. The transitions corresponding to absorbing
light could take place in the infrared on the right and
in the ultraviolet on the left, each of which would
result in a colorless appearance. You cannot
determine the crystal field strength from the information
given.
( 4 points ) |
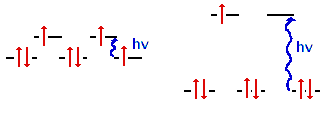 |
|
The magnetic properties of the
complex ion mentioned above are found to be identical to those of
the complex ion of V2+ with carbon monoxide ligands, but it is not
known if that complex is octahedral or tetrahedral. Explain
briefly what I can be learned from this information, if anything.
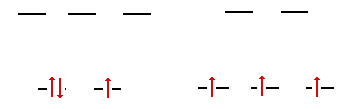
V2+ has 21 electrons and a
configuration given by [Ar]3d3 . Since carbon monoxide
is a strong field ligand, the configuration of the vanadium ion
in an tetrahedral field and a octahedral field would be as shown
below.
The conclusion you should
draw is that if this vanadium complex is tetrahedral,
then the "Bettis" ligand is a strong field
ligand, whereas if the vanadium complex should be
determined to be octahedral, then this would imply that
the "Bettis" ligand is a weak field ligand
according to the possibilities in the first part.
( 6 points ) |
 |
|