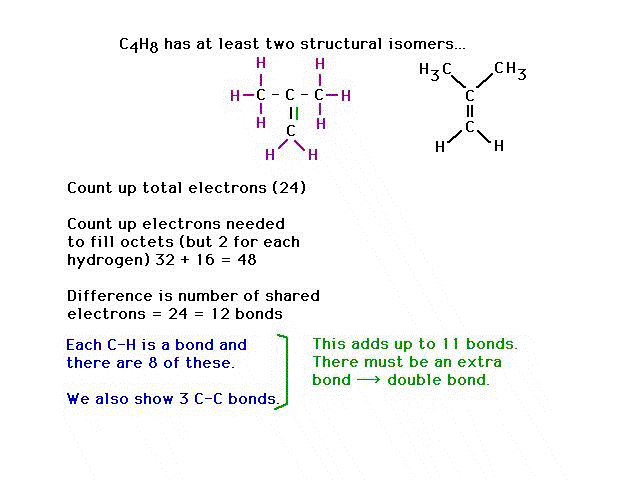
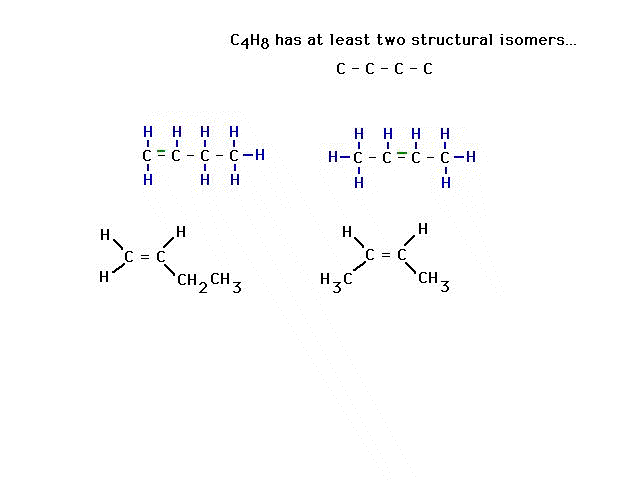
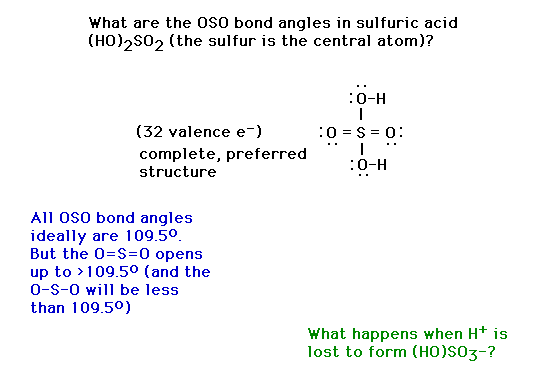
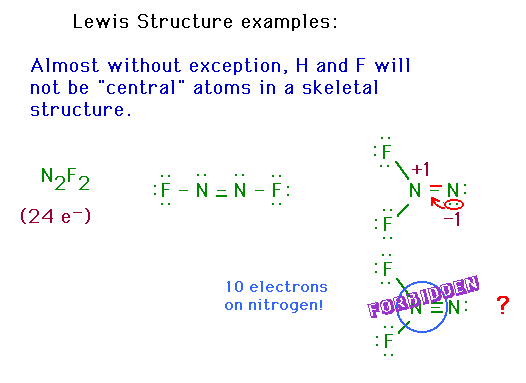
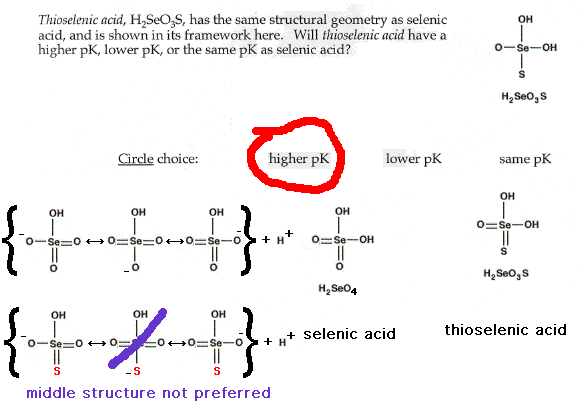
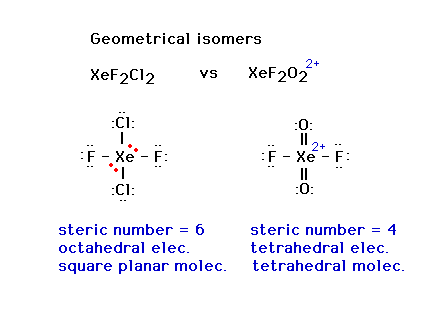
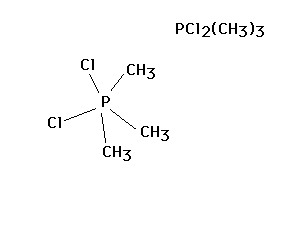
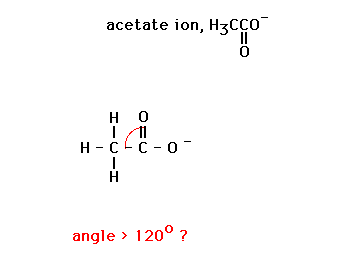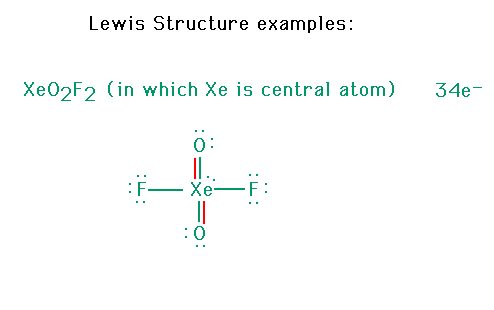| CURMUDGEON GENERAL'S WARNING. These "slides" represent highlights from lecture and are neither complete nor meant to replace lecture. It is advised not to use these as a reliable means to replace missed lecture material. Do so at risk to healthy academic performance in 09-105. |


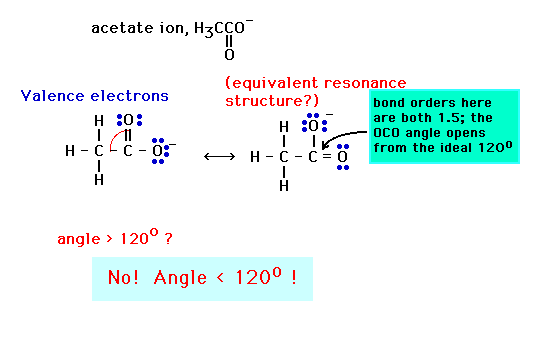
In the first, all angles are 120o since the benzene ring, because of resonance, is a perfect hexagon with 120o internal angles.
The second, after focussing attention on the N as a central atom has its lone pair compressing the other single bonds closer than their ideal 109.5o (despite having drawn the lone pair as if it were inside the ring -- which it isn't)